APPLICATIONS
CONFIDENCE WHEN YOU NEED IT MOST
-
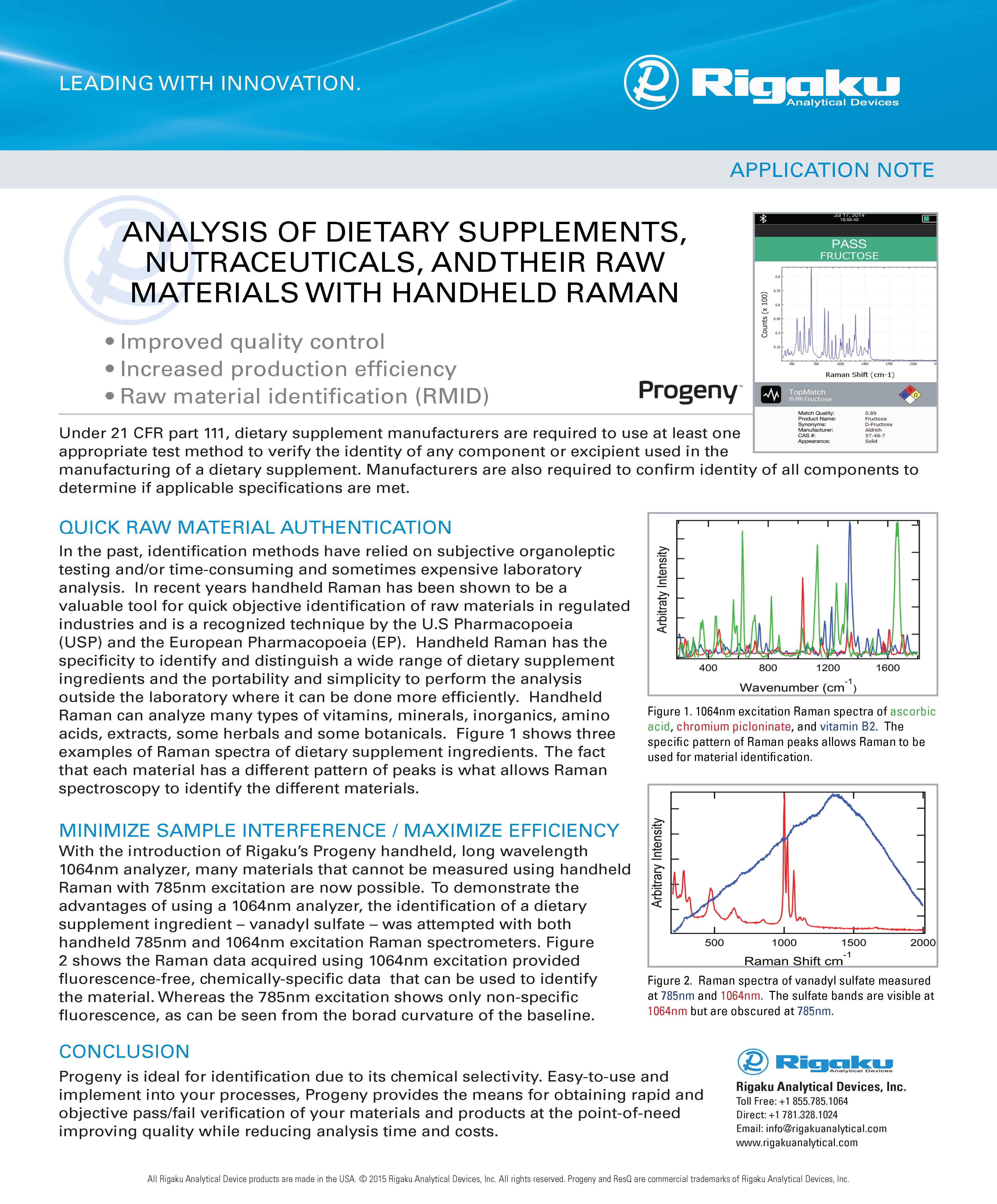
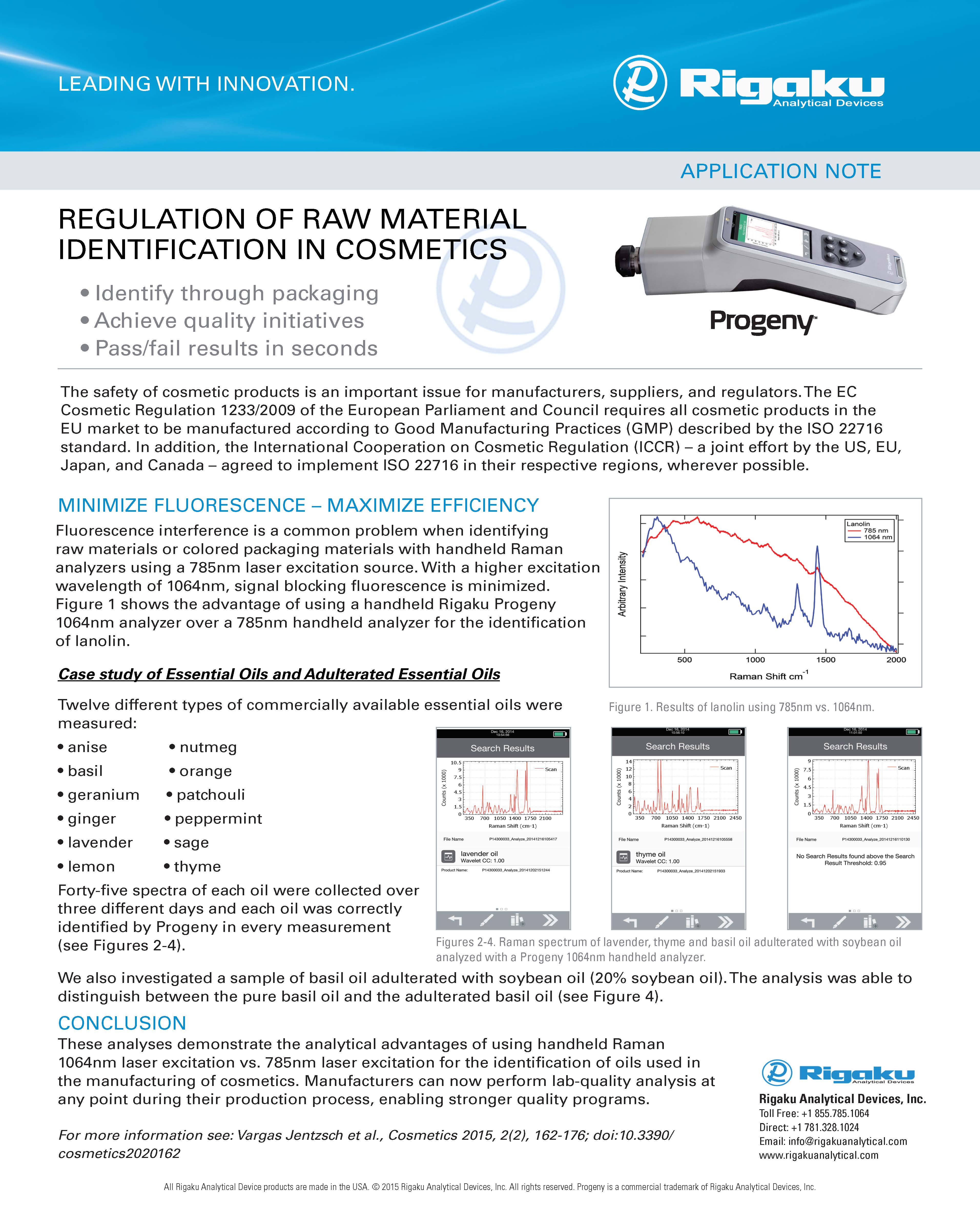
Raw material identification (RMID) of pharmaceutical, nutraceutical and cosmetic ingredients
-
Verification of trial materials, chemical, solvents and cell culture media
-
Authentication of finished products and anti-counterfeit prevention
HAND SANITIZER ANALYSIS
LEARN MORECOSMETICS
LEARN MORECELL CULTURE MEDIA
LEARN MOREPOLYSORBATES
LEARN MORECOUNTERFEIT MEDICINES
LEARN MORENUTRACEUTICAS / DIETARY SUPPLEMENTS
LEARN MOREEXCIPIENTS
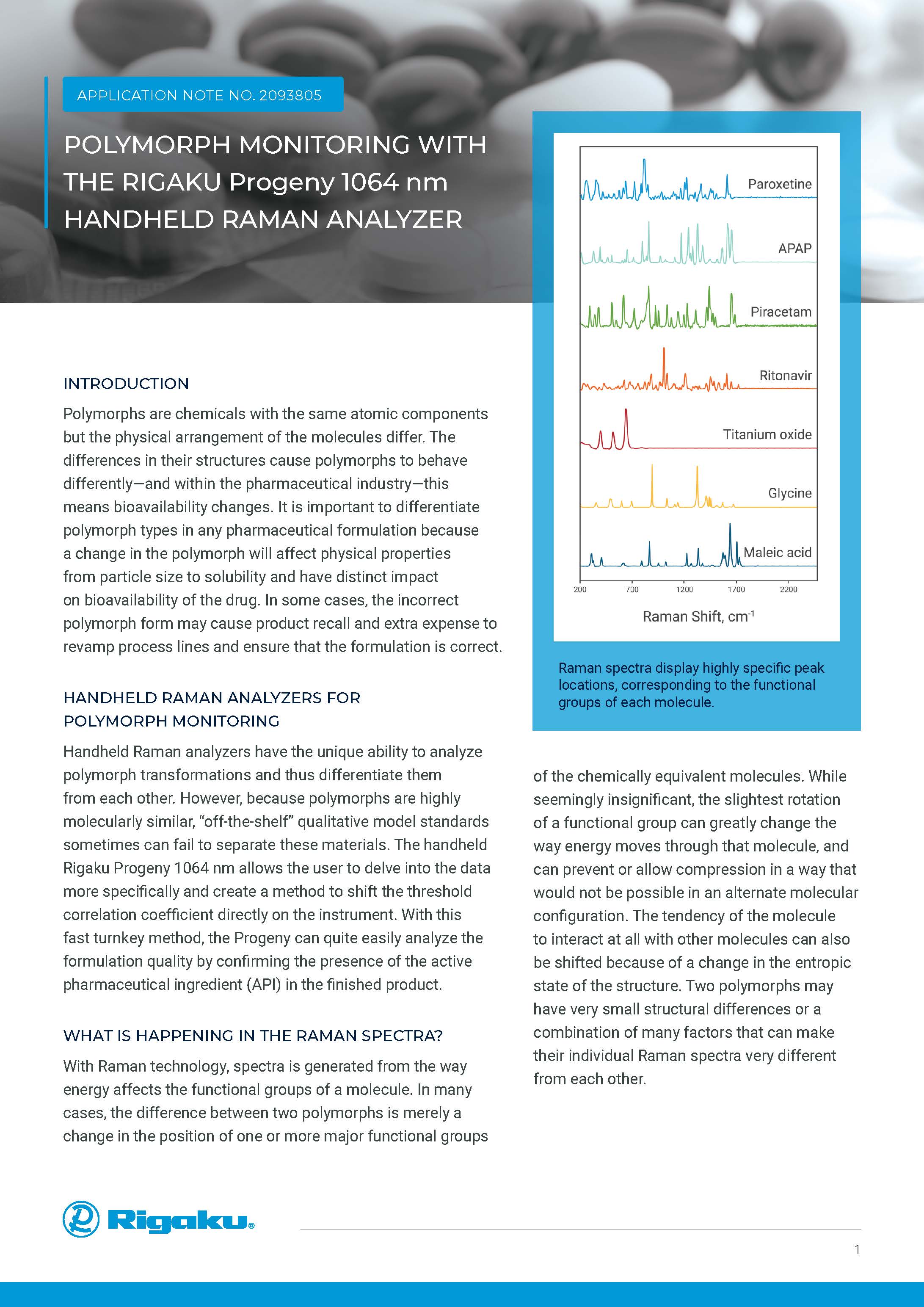
LEARN MOREPOLYMORPH MONITORING
LEARN MOREINDUSTRY-LEADING SUPPORT

"We use Progeny at every point in our manufacturing."

"Another thing I like about Progeny is the software as it relates to data integrity, ALCOA and those type of principals that you’re operating under a GMP environment.”

“I came at this with very little analytical background…and I found the instrument really easy to use…”

"The fact that we can continue to add new materials to the library is a huge benefit and the device is now also being used to establish ID methods in new product ingredients."
OUR COMMITMENT TO CUSTOMERS
- 24/7 PHONE SUPPORT
- IQ/OQ/PQ PROGRAM
- SOP DEVELOPMENT
MADE IN THE USA
FOCUSED CUSTOMER SUPPORT
TRUSTED APPLICATIONS
QUALITY CONTROL OF API POTENCY, EXCIPIENT BLEND UNIFORMITY, AND HEAVY METALS IMPURITIES BY NON-DESTRUCTIVE AND DIRECT ANALYSIS OF INTACT PILLS BY XRF
DIRECT CNOX ANALYSIS OF SOLID FORMS FOR CONFIRMATION OF COMPOSITION AND DETERMINATION OF SALTS/STOICHIOMETRY, HYDRATES, AND SOLVATES BY WD-XRF
THE USE OF EASY XRF ELEMENTAL ANALYSES TECHNIQUES TO SUPPORT THE DEVELOPMENT OF FORMULATIONS
A NON-DESTRUCTIVE XRF TECHNIQUE FOR RAPID AND EASY SCREENING OF RESIDUAL CATALYSTS IN API'S AND INTERMEDIATES
XRF WORKSHOP-BUILDING QUANTITATIVE XRF METHODS ON THE ZSX PRIMUS IV
FEATURES & BENEFITS
- Intelligent sequential wavelength dispersive X-ray fluorescence (WDXRF) for pharmaceuticals.
- Can be used for liquids, powders, and solids.
- Tube above or tube below configurations using a 4kW source with thin 30μm window gives enhanced light element performance ideal for ultra-light element analyses
- Walkup instrument (powder or pill in a cup) for routine high sensitivity elemental analysis in minutes.
- Rapid heavy-metal catalyst impurity analysis for process chemistry screening as well as metal impurities analyses according to ICH Q3D (USP 232 / 233 with detection limits sub PPM for class 1 elements (Cd, Pb, As, Hg).
- Results comparable with ICP and works well as a synergistic triage measurement prior to more extensive ICP analysis.
- Intact pills can be non-destructively analyzed for API potency, excipient blend uniformity and heavy metal impurities.
- Direct Carbon, Nitrogen, Oxygen (CNO-X) quantitation for confirmation of molecular formula and stoichiometry of salts, co-crystals, hydrates and solvates.
- Micro-analysis, contaminant analysis and elemental mapping with X-ray spot size down to 500μm.
- Quantitative analysis methods can be built using a combination of physical standards and standardless sensitivity libraries giving enhanced flexibility in method development.
TRUSTED PARTNER
Pharmaceuticals have the power to change the world for the better, but before they can ever do that, they need to be proven safe and trustworthy. Here at Rigaku, we strive to make this a reality as the leading global scientific analytical instrumentation company specializing in X-ray and thermal analysis, and Raman spectroscopy.
CORPORATE MISSION
To contribute to the enhancement of humanity through scientific and technological development.
CORPORATE MOTTO
Value our customers, value our people, and value our technology
HOW WE WORK WITH YOU
1 - FREE CONSULTATION
A CALL TO UNDERSTAND YOUR CHALLENGES
NEED TO REDUCE COSTS?
NEED A SPECIFIC APPLICATION?
HOW CAN WE HELP YOU?
2 - GET A DEMO
SCHEDULE A DEMO ON YOUR TERMS
ON-DEMAND RECORDINGS
LIVE VIRTUAL & CUSTOM
IN-PERSON GROUPS & INDIVIDUAL
3 - GET A QUOTE
BUILD SOLUTIONS PACKAGE RIGHT FOR YOUR NEEDS
TECHNOLOGY
INSTALL & TRAINING
SUPPORT & SERVICE
RESOURCES
- BROCHURE
- SPECIFICATION SHEET
- APPLICATION NOTES
- FAST FACTS
- WHITE PAPERS
- CASE STUDIES
- ACCESSORIES






_Page_1.jpg)